|
7/5/2023 0 Comments Pf3 molecular geometry In the figure below, you can see that the central electron has an octet because it has six particles. However, we don’t need to understand it every time as these rules are rare and happen only in extreme circumstances.Įxception 1 : If the octet has an odd number of valence electrons like 3,5,7, etc.Įxception 2 : If the octet has very few valence electronsĮxception 3 : If the strict has too many valence electrons. 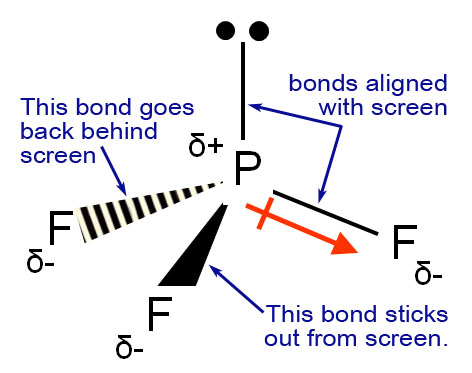
One thing to remember is that while sketching Lewis’s structures, the Octet Rules can be broken in these three scenarios. Therefore, there are no extra electrons (24 minus 24 equals 0). And additional electrons for the central atom, now add octets and extra electrons. There are 24 electrons here, and then we should add octets of the outer atom. To draw a Lewis Structure, you need to start adding electrons and connecting them. Even though Boron only has six valence electrons, we can see that the Boron Trifluoride Lewis structure’s formal charges are zero. Boron will be the least electronegative element at the core of its structure, and its outer shell also needs six valence electrons. You also need to count the number of valence electrons in Boron Trifluoride and then position them correctly before completing the octets. The central atom can be BF 3 which has 24 valence electrons, which must be rearranged around it. BF3 Lewis Structureįor determining the lewis structure, you need to calculate the total number of valence electrons for the BF 3 molecule. There are 120° bond angles on each of them that make them an equilateral triangle. 
This plane seems like all peripheral atoms exist in one place. This model contains three atoms that wrap around one atom in the centre and is referred to as a ‘Trigonal Planar’. 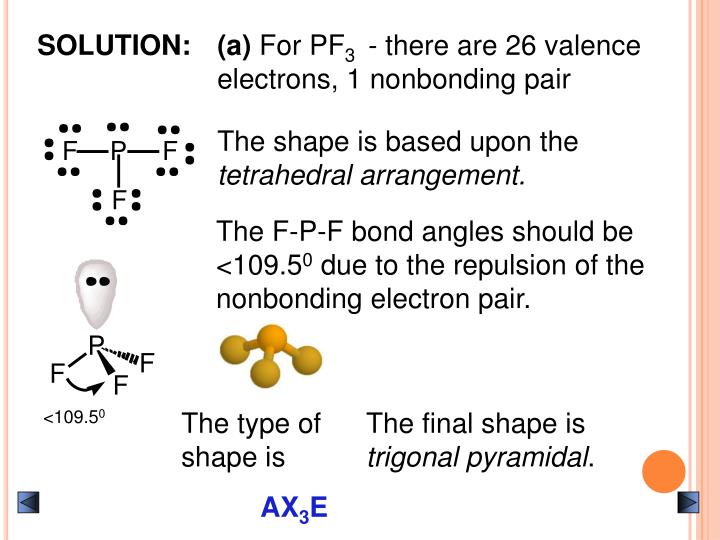
The BF3 molecule exhibits a ‘Trigonal Planar’ geometry. In damp air, it releases white vapours and is soluble if it is in the form of a colourless liquid (i.e., dihydrate.) Molecular Geometry of BF3 BF 3 is colourless, poisonous gas that has no colour. The inorganic compound is boron trifluoride with formula BF 3. These compounds are known as “inorganic compounds.” These compounds do not have carbon and are not called not organic. You may have heard of things like chemical molecules that don’t have any C-H bonds. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
